Is the 810 nm or 1064 nm (1070 nm) wavelength better?
Across all age groups, the 810nm wavelength has shown to have a deeper and stronger energy disposition than 1064 nm (1070 nm) in a dosimetry study by Harvard Medical School, Department of Psychiatry and several other universities. Even though 1064 nm (1070 nm) scatters less, it is absorbed more by water molecules, which are abundant in human tissue, especially the brain (70-80% water).
In terms of cellular effects, 810 nm has a stronger effect on mitochondria because photonic absorption by cytochrome c oxidase (CCO) peaks around 810nm and declines as the wavelength gets longer. Direct CCO photoexcitation is weaker at 1064 nm and 1070 nm compared to 810 nm because they are off-peak for mitochondria’s CCO absorption, which peaks around 810 nm.
On the other hand, 1064 nm (1070 nm) has a stronger effect on calcium ion channels, which 810 nm does not have a strong effect on.
The rest of this article, complete with science references, expands more on the differences, covering well-studied biophysics-based biological effects.
Does 810 nm or 1064 nm (1070nm) penetrate deeper into the brain?
According to a transcranial brain photobiomodulation (PBM) study by Harvard Medical School, Department of Psychiatry, the 810nm wavelength has been found to be superior to other wavelengths, which includes higher wavelengths in the 1070nm range for penetration and dosimetry.
According to this study by Harvard Medical School, the order of penetration and dosimetry effectiveness is:
-
810 nm – consistently highest across all age groups and regions
-
850 nm and 1064 nm – next most effective in most cases
-
670 nm and 980 nm – lesser deposition overall
This Harvard study is also supported by another brain PBM dosimetry study by leading Chinese universities, comparing 660 nm, 810 nm, 880 nm and 1064 nm. They discovered that the distribution of photon fluence at 660 and 810 nm within the brain was much wider and deeper than 980 and 1064 nm.

The distribution of photon fluence at 660 nm, 810 nm, 980 nm and 1064 nm. Wang P, Li T. “Which wavelength is optimal for transcranial low-level laser stimulation?” J. Biophotonics. 2019; 12:e201800173. https://doi.org/10.1002/jbio.201800173
The differences in dosimetry are supported by a well-established biological principle, the body’s first optical window. While, the 1064 and 1070nm wavelengths are longer and scatter less, they are more strongly absorbed by water, which is abundant in biological tissues, especially the human. The brain consists of 70-80% water, and floats in cerebrospinal fluid (CSF) while the rest of the human body is approximately 60% water. This makes wavelengths like 1064 nm and 1070 nm particularly susceptible to water absorption within the brain.
This increased absorption by water can lead to reduced photonic availability and tissue penetration despite the longer wavelength, which the Harvard Medical study and Peking Medical University study reveal. These studies indicate that 810nm has a higher dosimetry than 1064 nm and by extension, 1070 nm.

The near infrared window or body’s optical window. Image source: Wang, Erica & Kaur, Ramanjot & Fierro, Manuel & Austin, Evan & Jones, Linda & Jagdeo, Jared. (2019). Safety and penetration of light into the brain. 10.1016/B978-0-12-815305-5.00005-1.
- Water Absorption: Light absorption by water increases significantly beyond ~950 nm, and water is abundant in biological tissue. At 1064 nm, absorption by water becomes substantial, which attenuates the light more than at 810 nm. This increased absorption reduces the effective depth of penetration, especially for energy reaching specific chromophores like cytochrome c oxidase (CCO).
- Cytochrome c Oxidase (CCO) Absorption: Mitochondria’s CCO’s absorption spectrum peaks around 810 nm, with a notable decrease in absorption beyond 1000 nm. This means that 810 nm light is more readily absorbed by CCO compared to 1070 nm.
Visual Proof: Near-Infrared Light Penetrating the Skull with Vielight Neuro 4
The Vielight Neuro delivers the deepest tissue penetration among brain photobiomodulation devices. In the demonstration video below with the Vielight Neuro, 810 nm near-infrared light—emitted at an irradiance of 250 mW/cm²—can be clearly seen penetrating through the calvaria of a real human skull. This highlights the exceptional transcranial performance of the Vielight Neuro and validates the wavelength’s well-documented ability to reach cortical tissue.
The Vielight Neuro features proprietary Vie-LED technology—highly specialized, custom-engineered LEDs designed to deliver optimal irradiance for brain stimulation without producing excess heat. To ensure safety and efficiency, we’ve intentionally limited the device’s power density to 50% of its maximum potential output. Even still, it features the highest irradiance in the field of brain photobiomodulation according to independent 3rd party tests.
Differences in cellular effects between 810nm and 1070nm
810nm has a stronger effect on mitochondria, cytochrome C oxidase (CCO)
The 810nm wavelength is well-known for its strong interaction with cytochrome c oxidase (CCO), a key enzyme in the mitochondrial respiratory chain. By enhancing the activity of CCO, the 810nm wavelength increases ATP production, reduces oxidative stress, and modulates reactive oxygen species (ROS). These effects are crucial for cellular energy metabolism, neuroprotection, and the promotion of cell survival.
1064 nm and 1070nm has a stronger effect on heat-sensitive ion channels
On the other hand, wavelengths beyond 900nm, such as the 1064nm and 1070nm wavelengths have a weaker effect on mitochondrial CCO but a more direct effect on heat-sensitive ion channels, due to its potential to cause localized heating. Activation of these channels can lead to increased calcium influx, which is crucial for various cellular processes, including neurotransmitter release, gene expression, and neurogenesis.

The effects of red to NIR light energy on mitochondria Ref: Original: “Basic Photomedicine”, Ying-Ying Huang, Pawel Mroz and Michael R. Hamblin, Harvard Medical School. Current design: Vielight In
Mitochondrial Activation
- 810nm Wavelength:
The 810nm wavelength is particularly effective in targeting cytochrome c oxidase, which is a critical component of the mitochondrial electron transport chain. This wavelength is more efficiently absorbed by cytochrome c oxidase, leading to a robust activation of the mitochondrial respiration process. As a result, there is an increase in ATP production, which supplies energy to cells and supports various cellular functions. The 810nm wavelength is especially effective in reaching superficial and cortical brain regions, promoting enhanced cellular metabolism and function in these areas.[2]
- 1064 nm and 1070nm Wavelengths:
The 1064 nm and 1070 nm wavelengths, while still within the near-infrared (NIR) spectrum, does not interact with cytochrome c oxidase as effectively as the 810nm wavelength. The absorption by mitochondrial chromophores decreases significantly as the wavelength increases beyond 810nm.[5] Consequently, the 1070nm wavelength has a reduced effect on mitochondrial activation when compared to 810nm. Instead, the 1070nm wavelength might exert its effects through other mechanisms, such as potential thermal effects and heat/light ion gated channels.
Neurogenesis
- 810 nm Wavelength:
At the cellular level, the 810nm wavelength has shown considerable efficacy in promoting cortical neurogenesis—the process by which new neurons are formed in the brain. This wavelength is also known for its anti-inflammatory effects, which can help reduce neuroinflammation and support the brain’s healing processes. The 810nm wavelength is well-suited for applications targeting the outer layers of the brain, where it can stimulate cellular repair mechanisms, reduce oxidative stress, and promote overall Brain wellness.
Flowchart of Differences in Cellular Mechanisms:
-
1064/1070 nm → water-mediated microheating → TRP gating/membrane-capacitance effects → Ca²⁺ influx.
-
810 nm → CCO-mediated mitochondrial signaling → downstream Ca²⁺ effects.
Why mitochondria absorbs 810 nm more than 1064 nm (1070nm)
1. Spectral absorption properties of cytochrome c oxidase (CCO) within mitochondria
-
Cytochrome c oxidase within mitochondria has distinct absorption bands in the visible red (~660 nm) and near-infrared (810 nm) regions. Studies consistently show that absorption drops off as you shift to longer NIR wavelengths around the 1000 nm range like 1064 nm (1070nm).
-
The absorption bands of CCO become much weaker at wavelengths greater than 900 nm, which suggests that alternative chromophores must exist significantly.
2. Reduced photon availability at 1064 nm (1070 nm)
-
At ~1064 nm (1070nm), water (the dominant tissue component) is absorbed significantly versus 810 nm. This leads to greater attenuation (loss) of photons before they can reach and excite CCO.
Why calcium ion channels absorb more 1064 nm (1070nm) versus 810 nm
Calcium channels themselves don’t meaningfully “absorb” 1064 nm light. What happens at ~1064–1070 nm is mainly photothermal coupling – water is absorbed significantly at longer NIR wavelengths (> 1000 nm), producing tiny, rapid temperature rises that gate heat-sensitive TRP calcium channels (e.g., TRPV1/2/4) and/or change membrane capacitance, which in turn drives Ca²⁺ influx.
By contrast, 810 nm couples primarily to cytochrome-c-oxidase (CCO) with much weaker water heating, so Ca²⁺ effects there are usually downstream of mitochondrial signaling, not direct channel gating.
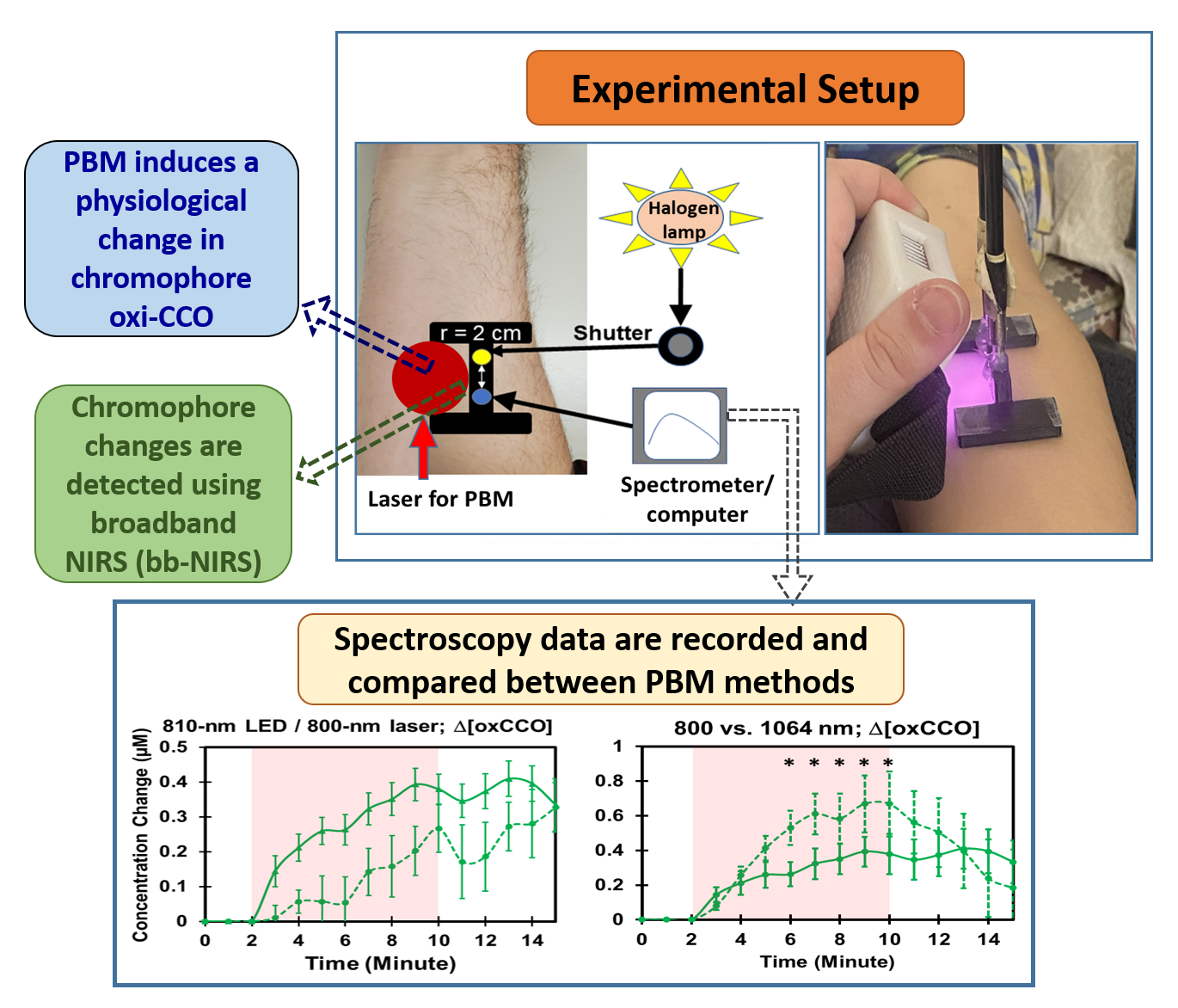
Research validation of 810nm LEDs vs lasers
A recent study on vascular hemodynamics and cytochrome c oxidase redox activity (not on the brain, but on arms) by the Department of Bioengineering, University of Texas at Arlington examined the effects of different wavelengths within this range with
- Lasers (800-1064nm | 250 mW/cm2 )
- 810nm LED (135 mW/cm2)
Results
- The 810 nm LED was able to create significant stimulations on vascular hemodynamic oxygenation and CCO redox metabolism despite the LED having a lower irradiance (≈135 mW/cm2)
- The dose-dependent trajectory by the 810 nm LED was similar to that by the 800 nm laser.
- The LED-triggered increases in Δ[oxCCO] remained at the elevated level without a returning tendency at least during the 5 min post-PBM period. In contrast, the increased Δ[oxCCO] by the 1064 nm laser started returning to the baseline immediately after the cease of the laser.
These findings are encouraging for us – our Vielight Neuro’s rear 810nm LED transcranial diodes generate ≈200-300 mW/cm2, which surpasses the power density of ≈135 mW/cm2 used in the study. It underscores our commitment to fewer well-placed but sufficiently powerful diodes vs many weaker diodes.
An important takeaway is the importance of irradiance values (mW/cm2 ) in this study.
Wavelength alone isn’t enough – irradiance matters.
Irradiance—also referred to as power density or light intensity – is a measure of how much light energy reaches a surface per unit area, typically expressed in milliwatts per square centimeter (mW/cm²). In photobiomodulation (PBM), including brain PBM, irradiance determines how much photonic power is delivered to the tissue.
While using an effective wavelength (such as 810 nm, 1064 nm, or 1070 nm) is essential for targeting chromophores like cytochrome c oxidase, the biological response and penetration depth depends just as much on irradiance. Without sufficient power density, even the correct wavelength may fail to penetrate tissue effectively or trigger meaningful cellular effects. Low irradiance can result in sub-therapeutic penetration and doses, while excessively high irradiance may lead to phototoxicity or energy wastage.
In a published review study of over 2133 brain photobiomodulation studies, from which 97 studies were included, the average irradiance or power density was around 250 mW/cm2
As a point of comparison, the average surface irradiance of near-infrared (NIR) light in natural sunlight is approximately 45 mW/cm² – a helpful benchmark when evaluating PBM device output.
In short, optimal photobiomodulation requires both the right wavelength and the right irradiance to reach the target tissue and activate mitochondrial responses.
Key Concepts:
- The NIR spectrum of sunlight has an average irradiance or surface power density of 45 mW/cm²
- Afternoon sunlight is free. To provide a meaningful therapeutic advantage, brain photobiomodulation devices must deliver higher irradiance levels than the NIR range in sunlight, ensuring benefits beyond what can be achieved through standard sunlight exposure.
- Sunlight contains harmful UV rays within 100-400nm range. Brain photobiomodulation devices only emit beneficial light energy within the 810-1100nm range.
A Comparative Snapshot
In a 2024 systematic review that screened 2,133 records and included 97 brain-PBM studies, reported power densities typically clustered around ~250 mW/cm² (especially under physiological conditions).
This is a snapshot comparison of independently measured irradiance by photonics labs by the PBM Foundation between commercial devices with the 810nm wavelength and the 1064 nm, 1070nm wavelengths:
Irradiance / Power Density Comparison
Vie-LED technology is unique and is engineered to generate a laser-like irradiance profile but with the safety of LEDs.
The PBM Foundation benchmarked the Vielight Neuro 3 against two PBM helmets, the Suyzeko NIR helmet and Neuronic Neuradiant twice, as case studies for their testing program to standardize irradiance reporting.
MegaLab and Optronic Lab, photonics engineering firms, conducted the tests:
When compared against the irradiance of peak natural sunlight (which is free) our Vielight Neuro generates 200-300% the irradiance of sunlight without the negative side effects of UV rays. The tested Neuronic and Suyzeko helmets generated less than 12% of sunlight’s peak irradiance.
A 2024 systematic review that screened 2,133 records and included 97 brain PBM studies reports that irradiance (power density) was typically ~250 mW/cm². Which implies that the Neuronic and Suzyeko helmets generated less than 5% of the average irradiance analyzed over 97 brain PBM studies. The Vielight Neuro slightly exceeds the irradiance used in these studies, which included lasers.
| Source | Independently measured irradiance | Manufacturer | % of Typical Brain-PBM Irradiance (≈250 mW/cm²) |
|---|---|---|---|
| Vielight Neuro (Vielight) | 180-350 mW/cm2 | Vielight, Canada | 80–160% |
| Neuradiant 1070 (Neuronic) | ≈9 mW/cm2 | Suyzeko, China (Private-labelled) |
≈4% |
| Suyzeko PBM Helmet (Suyzeko) | 5 mW/cm2 | Suyzeko, China | 3% |
| Natural Sunlight | 100 mW/cm2 | Free | 40% |
Number of published clinical studies
Vielight technology is featured in the most published research by a significant margin for the reasons above.
Be cautious of companies attributing research conducted with Vielight devices or other devices as attainable to their own.
Brain photobiomodulation is parameter-specific and our Vie-LED technology generates a unique laser-like profile and an industry-leading irradiance.
The table below is a benchmark studies published comparison against other random PBM helmets.
| Technology | Independently measured wavelength | Research | Manufacturer | Medical Grade |
|---|---|---|---|---|
| Vielight Neuro (Vielight) | 810nm | 20 published (17 ongoing) |
Vielight, Canada | Yes |
| Neuradiant 1070 (Neuronic) | 1059nm | 2 published | Suyzeko, China (Private-labelled) |
No |
| Suyzeko PBM Helmet (Suyzeko) | 811nm | 1 published | Suyzeko, China | No |
Conclusion
Both 810 nm and 1070 nm wavelengths are widely used in brain photobiomodulation (PBM), and emerging evidence suggests each has distinct advantages depending on the clinical context. Numerous independent studies have focused on 810 nm, with consistent positive outcomes in mitochondrial activation, neuroprotection, and cognitive benefits. In contrast, research using the 1070 nm range—such as 1064 or 1070 nm—though less abundant, shows similar therapeutic potential and quality when conducted.
Vielight’s selection of 810 nm is rooted in its lower absorption by hemoglobin and water, enabling deeper and more efficient penetration through scalp, skull, and brain tissue than higher wavelengths like 1070 nm. Recent peer-reviewed comparisons—including work by Harvard Medical School—confirm that 810 nm reaches deeper brain structures under comparable power density conditions.
While 1064 nm and 1070 nm may offer slightly better photon scattering properties for deep tissue delivery, especially in neurological conditions, mitochondrial stimulation efficacy tends to be stronger with 810 nm, owing to its specific absorption by cytochrome c oxidase and related chromophores. That means while both wavelengths are effective, 810 nm is often seen as optimal for combining depth with bioenergetic stimulation, whereas 1064 nm 1070 nm is a reasonable alternative for the effects on calcium ions.
However, both 810 nm and 1070 nm wavelengths require strong irradiance levels to achieve therapeutic efficacy, particularly when targeting brain tissue. This is because transcranial photobiomodulation must overcome several biological barriers—including the scalp, skull, and cerebrospinal fluid—before sufficient light can reach neuronal structures. Higher irradiance (measured in mW/cm²) ensures that enough photons penetrate these layers and maintain adequate energy density at depth to activate key chromophores such as cytochrome c oxidase. Without sufficient irradiance, even an optimally chosen wavelength may fail to deliver meaningful biological effects.